How I Explain Inspire to My Patients

Inspire is like a pacemaker for your breathing. It’s placed through two small incisions — one in your neck and one in your chest, both on the right side. Through the neck incision, we place a small stimulation cuff around the nerve that controls your tongue. We then tunnel a wire down to the chest, where it connects to the battery that powers the device.
The battery lasts about eleven years and never needs to be recharged. The surgery takes roughly an hour, and it’s outpatient — you go home the same day.
About a month later, once you’re fully healed, we activate the device in the office. You’re given a small remote and taught how to use it. At bedtime, you press a button to turn the device on. It pulses once to let you know it’s working, then delays about thirty minutes so you can fall asleep.
Once the therapy kicks in, Inspire senses your breathing with every breath and gently pulses your tongue forward to keep your airway open. This typically reduces sleep apnea by about 70%. You’ll come in about twice a year to have the device checked, and we’ll do a home sleep study three to four months after surgery to confirm everything is working well.
What’s Actually Being Implanted?
The current generation of Inspire (Inspire V) has just two components, all hidden beneath the skin.
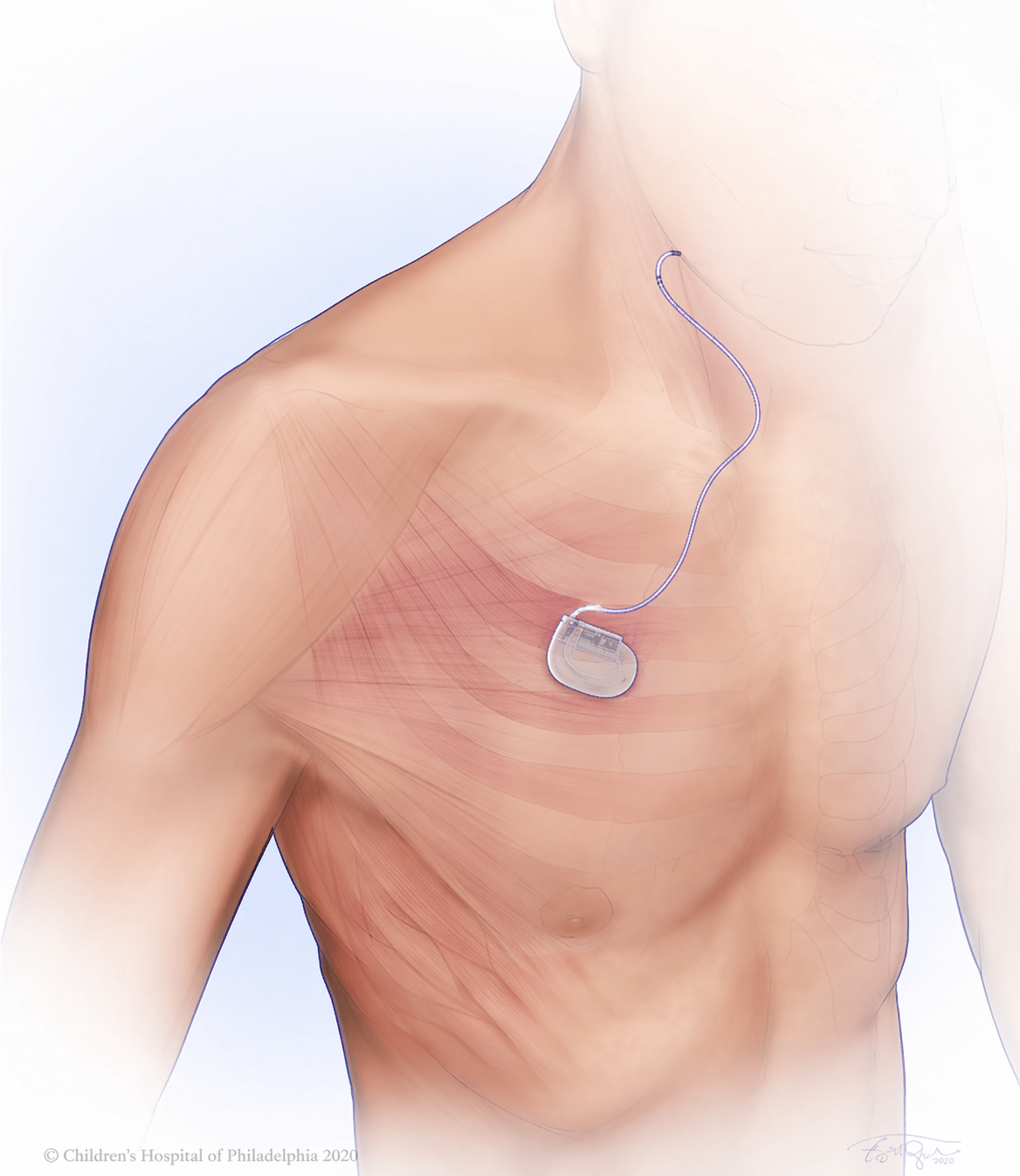
What Does Recovery Look Like?
Most patients are surprised by how manageable the recovery is. The device stays off during the entire healing period — there's no rush.
Am I a Candidate?
Candidacy depends on your sleep study results, airway anatomy, and what your insurance requires. Use the criteria below as a starting point — then check the coverage guide to see how they apply to you.
Insurance criteria vary by plan — deductibles, BMI cutoffs, and CPAP-trial requirements can all change what you qualify for.
See if you might qualify →What the Data Shows
Inspire is one of the most studied surgical treatments for obstructive sleep apnea. The landmark STAR trial was published in the New England Journal of Medicine in 2014, with results sustained through four-plus years of follow-up.
Apnea Reduction
Patients went from an average AHI of 29 down to about 9 at twelve months — a 68% reduction in the number of times the airway closes per hour of sleep. By three years, the average AHI had improved further to under 5.
Snoring
Before surgery, only 18% of bed partners reported soft or no snoring. After twelve months with Inspire, that jumped to 85%. The percentage of bed partners who had to leave the bedroom dropped from 30% down to 5%.
Daytime Sleepiness
Patients’ Epworth Sleepiness Scale scores dropped from 11 (abnormally sleepy) to 6 (normal range) — and stayed there through four years of follow-up.
Quality of Life
Functional quality-of-life scores normalized after surgery and remained stable long-term. 63% of patients reported a clinically meaningful improvement in daily functioning. Over 41,000 patients have been implanted worldwide.
How Inspire Compares to Traditional Sleep Surgery
Inspire doesn’t change your anatomy — it works with your anatomy. Unlike traditional tissue-removing surgeries, the therapy is adjustable and reversible.
Living with Inspire
Once you’re up and running, the nightly routine is simple. Press a button at bedtime. Turn it off in the morning. That’s it.
This page is for educational purposes only and does not constitute medical advice. Individual results may vary. Clinical data referenced from the Inspire STAR Trial (Strollo et al., N Engl J Med, 2014) and the ADHERE post-approval registry. Inspire is an FDA-approved device for the treatment of moderate to severe obstructive sleep apnea in patients who meet specific eligibility criteria. Always consult with your physician to determine the best treatment option for your individual needs.
